- Dangerous Goods are articles or substances which are capable of posing a risk to health, safety, property or the environment and which are listed as such in the Technical Instructions or which are classified according to the Technical Instructions
- Carriage of Dangerous Goods by air poses an inherent safety hazard; because of the characteristics of air transport (vibration, pressure and temperature changes) can have such an effect of articles, substances and packaging to pose a risk to the aircraft, passenger and crew that may not be apparent on the ground. There are some items that pose such a risk at any time, whether they are being transported or not
- Dangerous Goods are commodities that possess potentially dangerous characteristics. These characteristics will not take their transport by air dangerous, provided that suitable precautions are taken. Dangerous Goods consist not only of obvious substances such as acids, explosives, and poisons but also include less apparent articles like magnets, refrigerants and some everyday household items such as bleaches, aerosols, and perfumes
- To ensure safe transport of Dangerous Goods by air, ICAO has laid down the standards and recommended practices in Annex 18. ICAO has also issued Technical Instructions for Safe Transport of Dangerous Goods by Air, the document (Doc 9284- AN/905) which gives classification, list of Dangerous Goods, packing, labeling, marking, documentation and training of concerned personnel and other associated aspects. In India, the Aircraft (Carriage of Dangerous Goods) Rules, 2003, have been framed by Director General of Civil Aviation to give effect to these provisions made by ICAO
- There are internationally agreed rules to ensure the safe transportation of dangerous good by air. These are published by the International Civil Aviation Organisation (ICAO) as the Technical Instructions for the Safe Transport of Dangerous Good by Air
- The IATA Dangerous Goods Regulation Manual (abbreviated DGR in this Manual) is based on Annex 18 to the Chicago Convention of International Civil Aviation Organization. They include all the requirements of Annex 18 and the latest ICAO
- Technical Instructions. The IATA Dangerous Goods Regulation Manual is recognised by ICAO as the field document for the transport of dangerous good by air
Indian Regal Regime for carriage of Dangerous Goods
The carriage of dangerous goods to/from/within/over India is governed by the following:
- The Aircraft Act 1934 - Section 5(2) (L) and Section 10(1)
- The Aircraft Rules 1937 - (Rule 8)
- The Aircraft (Carriage of Dangerous Goods) Rules, 2003
- Civil Aviation Requirements – Section 11 – Safe Transport of Dangerous Goods by Air Series ‘C’ Part I, - Issue III dated 08th January, 2010 (Guidelines and Procedure for Dangerous Goods Inspections)
- Civil Aviation Requirements – Section 11 – Safe Transport of Dangerous Goods by Air Series ‘C’ Part I, Issue III dated 08th January, 2010 (Approval of Dangerous Goods Training Programme)
Basis of International Dangerous Goods Regulation
- UNCOE gives recommendations and procedure for handling of dangerous goods other than Radioactive Materials. International Atomic Energy Agency (IAEA) recommends the procedure for handling Radioactive Material
- International Civil Aviation Organization (ICAO): ICAO uses these recommendations and ICAO Annex 18 to develop a Technical Instructions for Safe Transport of Dangerous by Air, which provides the basis under which dangerous goods can be transported safely by air
- IATA Dangerous Goods Regulations: Airline uses the IATA Dangerous Goods Regulations as everyday field documents. This document fully complies with ICAO Technical instructions & Annex 18 and some cases more restrictive by taking account of industry standard practices or operational considerations
- It is ensured that a current edition of the IATA Dangerous Goods Regulations (DGR), the ICAO Technical Instructions for the Safe Transport of Dangerous Goods by Air (Technical Instructions), along with this manual must be accessible at each location where ground handling operations involving the acceptance or loading of dangerous goods as cargo are conducted
Dangerous Goods carried in IndiGo
- Dry – ice as packing material
- Radioactive material (FDG 18)
Dry ice used as packing material (Proper Shipping name: Carbon dioxide, solid)
General
Dry ice (or carbon dioxide solid, UN 1845) is considered a dangerous good/hazardous material for air transport and requires special handling. Dry ice is classified by DOT (Department of Transportation) and IATA as a “miscellaneous” hazard, class 9.
In IndiGo, Dry ice is used as a coolant for storing perishable like chocolates, ice-creams, blood samples, medicines, stickers etc.
DRY ICE used as packing material (Shipping name: Carbon dioxide, solid)
General
Dry ice (or carbon dioxide solid, UN 1845) is considered a dangerous good/hazardous material for air transport and requires special handling. Dry ice is classified by DOT (Department of Transportation) and IATA as a 'miscellaneous' hazard, class 9.
At IndiGo, dry ice is used as a coolant for storing perishable such as chocolates, ice-creams, blood samples, medicines, stickers, etc.
Why is dry ice dangerous?
Dry ice is considered hazardous during transportation for three reasons:
- Explosion hazard: dry ice releases a large volume of carbon dioxide gas as it sublimates. If packaged in a container that does not allow for the release of the gas, it may explode, causing personal injury or property damage
- Suffocation hazard: a large volume of carbon dioxide gas emitted in a confined space may create an oxygen deficient atmosphere
- Contact hazard: dry ice is a cryogenic material that causes severe frostbite upon contact with skin
General requirements
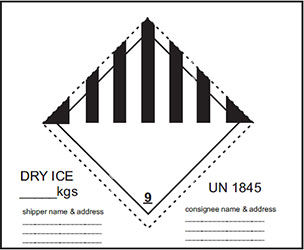
Dry Ice is a class 9 Dangerous Good. Packages containing dry ice must always be declared as such by proper marking and labelling. A ‘Shipper’s Declaration for Dangerous Goods’ is not required if no other dangerous goods are in the shipment but 'Instruction of dispatch of Goods' is required. Dry ice must be packaged to permit the release of carbon dioxide gas and to prevent a build-up of pressure that could rupture the packaging. Below Packing Instructions must be used and ‘dry ice checklist’ has to be filled by the Acceptance staff at the time of load being tendered. A sample of IndiGo Standard dry ice sticker is provided below:
Packing Instruction 954
- This instruction applies to UN 1845 on passenger and CarGo aircraft and CAO. The General Packing Requirements of section 5.0.2 of the current edition of the DGR must be met
- Carbon dioxide, solid (dry ice) in packages, when offered for transport by air, must be in packaging designed and constructed to permit the release of carbon dioxide gas and to prevent a build-up of pressure that could rupture the packaging. Arrangements between shipper and operator(s) must be made for each shipment, to ensure ventilation safety procedures are followed. The shipper must provide the operator with written documentation stating the total quantity of the dry ice contained in each box
- The Shipper's Declaration requirements of Subsections 8.1 and 10.8.1 of the DGR are only applicable when the Carbon dioxide, solid (dry ice) is used as a refrigerant for dangerous goods that require a Shipper's Declaration
- When a Shipper's Declaration is not required, the following information, as required by 8.2.3 of the DGR for the Carbon dioxide, solid (dry ice), should be contained in the ‘Nature and Quantity of Goods’box on the Air Waybill:
- UN 1845
- proper shipping name (Dry ice or Carbon dioxide, solid)
- Class 9
- the number of packages
- The net quantity of dry ice in each package
- The net weight of the Carbon dioxide, solid (dry ice) must be marked on the outside of the package
Dry ice used as a refrigerant for other than dangerous goods may be shipped in a unit load device or other types of pallet prepared by a single shipper provided that the shipper has made prior arrangements with the operator. In such a case, the unit load device, or other types of pallet must allow the venting of the carbon dioxide gas to prevent a dangerous build-up of pressure. The shipper must provide IndiGo with written documentation stating the total quantity of the dry ice contained in each packages of the consignment.
Special Instruction
- A maximum acceptable quantity of dry ice per piece will be 30 kg in case of Airbus 320 and 5 kg in case of ATR. The Max quantity per flight will be 200 kg for Airbus A – 320 and 10 kg for ATR72-600
- A letter from the shipper needs to be taken while accepting dry ice shipment stating the net quantity of dry ice per packet
- A pre-alert should be sent to Airport Operations, Transit station(s) and destination in a specified format
- The pilot needs to be issued a Special Load Notification (NOTOC). CarGo staff should prepare the NOTOC and hand it over to the Load and Trim staff for a signature of acceptance from the Pilot and one copy of the signed NOTOC has to be retained by CarGo staff for office records
- Loading in Airbus 320 holds:
- Hold-1: 100 kg max allowed
- Hold-3 & 4: 75 kg max allowed
- Hold-5: 25 Kg max allowed
No specific compartment – wise loading specification is given in IATA DGR
Things to note:
- Packages in which dry – ice is used as refrigerant is only acceptable
- Maximum amount of dry – ice per package is 30 kg
- Maximum amount of dry – ice per aircraft is 200 kg
- Boxes must perforate in such a way to allow cross – ventilation
Documents required for transportation of Shipment packed with dry – ice
- Completely filled Instruction for Dispatch of goods (IDG)
- Completely filled Security declaration
- Letter stating the quantity of Dry – ice in each package. (This is mandatory)
Packing, marking and labelling
- Legible marking on the packages are mandatory
- Emergency contact number to be clearly visible
- Specified Class 9 label (is available in the counter) to be affixed on each package
Radioactive Material (FDG)
General
Radioactive material means any material containing radionuclides where both the activity concentration and the total activity in the consignment exceed the values specified in 10.3.2 of the DGR manual.
IndiGo accepts only Fluorodeoxyglucose F-18 (FDG) as radioactive material (RRY) in its Airbus 320 aircraft, however, radioactive material is not accepted in ATR72 flights
Description of F-18 ( FDG)
Fluorodeoxyglucose, F-18 (FDG) is a radiopharmaceutical used in medical imaging with PET (positron emission tomography). This medicine is a radioactive drug FDG produced from Cyclotron and transferred to different locations (as per requirement). After FDG is injected into a patient, a PET scanner can form images of the distribution of FDG around the body that can be assessed to diagnose various medical conditions, cancer in particular. It can also be used to diagnose brain and cardiac disorders.
- Radioactivity: Half-life is 109.7 minutes. Will decay to normal radioactive background levels in 18 hours
- Health Hazards: Skin and eye irritants when exposed. Since small quantities present in each container, no adverse effects are expected to occur
- Fire /Explosion: No risk of fire or explosion
- UN number: UN 2915
- Proper Shipping Name: Radioactive material, Type A package,
- Hazard Class: 7
- Hazardous Polymerisation: No occurrence
- Physical State /Appearance: Clear, liquid in a glass vial
- Colour: Colourless
- Odour: Odourless
- Vapour pressure /Density: Not found
- Boiling Point: 100 oC (212 oF)
- Melting Point: 0oC (32oF)
- Solubility: Soluble in water
- Evaporation point: None
- Specific Gravity: >55Mbq/mg F18 – FDGPacking of the container
Packing of the container
The packaging instruction is given and approved by AERB (refer Certificate of Registration for Type A package design in the Atomic Energy Regulatory Board (AERB) Safety Code No. SC/TR – 1, 1986 edition for transportation of radioactive material). The packaging details are as follows:
Exterior Packaging: An external “Flight case” multiplex wood packaging
- Material: Plywood with stackable steel reinforced angles
- Weight: 7 kg 917.5 Gross wt
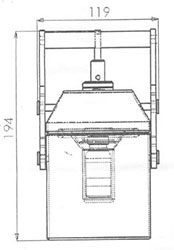
- Dimension: 280 X 280 X 330mm
A bracing system consisting of synthetic material and foam components to center and fix the lead container into the “flight case”.
Internal foam: Polyethylene high density 30 kg/cub M.
Container: Material: Lead container fully covered with stainless steel
- Weight: 10.5 kg
- Dimension: Dbody = 103mm; Dhandles = 119mm; H = 194mm
- Pb thickness = 32mm
- Stainless steel: 316 type 1.5mm electropolished, thickness 2mm
- Primary Container with lead shielding: 10ml glass vial capped with rubber bunk & aluminium cap loaded in the above shield container
- Lead container positioning: placed centrally
Health Hazard
Potential Health Effects: The hazardous, non – radioactive ingredients found in Fluorodeoxyglucose F 18 (FDG) injections are skin and eye irritants, but, because of the small quantities present in each container, no adverse effects are expected to occur from these components.
Eye Contact: No adverse effect expected, but may cause mechanical irritation. In high doses, may cause dryness of the eye or may lead to the development of cataracts.
Skin Contact: Not expected to produce any acute adverse health effects. High doses may cause erythema (skin burns).
Inhalation: FDG does not easily become airborne under standard conditions. Not expected to be a health hazard via inhalation.
May cause asymptomatic physiological uptake by specific target organs or tissues
Documents required to book FDG
Documents required to book FDG
The following documents are required from the shipper for transportation of RRY:
- Copy of the valid permission letter from Atomic Energy Regulatory Board (AERB) which allows them to transport FDG in a Type A container. This copy is mandatory for every shipment. (AERB is the ultimate authority to oversee manufacture and supply of radiopharmaceuticals. They give the license to the shipper for transporting radioactive material. The organisation provides the guideline for safe transportation of radioactive material)
- At least two copies in original of Shipper’s Declaration to Dangerous Goods as per section 8.1.2.3.1 of the current edition of the DGR manual
- Consignor’s Declaration for Safe Transportation of RRY
Shipment having sum of the Transport Index maximum up to 3 is accepted.
The following documents required to be forwarded to the destination
- A copy of the Airwaybill
- A copy of the DGD
- A copy of the AERB permission letter
- A copy of Consignor’s Declaration for Safe Transportation of Radioactive material
Acceptance procedure
- Acceptance will be performed latest by D – 90 mins. Prior to the departure of the flight
- Prior booking to be made in IndiGo CarGo booking system
- Radioactive material to be booked in the direct flights only
- Radioactive Checklist should be filled by DG CAT 6 staff
- Documentation as stated in section 1.2.1 to be in strict compliance
- Marking and labelling (Category II – yellow / Category III – the yellow label is used) to be checked. The responsibility of affixing label is to be affixed by the consignor
Emergency First Aid
Eye contact: Immediately rinse the eye with saline solution several times. Notify radiation safety personnel.
Skin Contact: If skin contact occurs, immediately wash the affected area thoroughly and vigorously with soap and water until the radioactivity can be removed. Always blot dry. Do not abrade the skin. Notify radiation safety personnel.
Inhalation: Not expected to require first aid measures.
Ingestion: Not expected to ensure first aid measures.
Other First Aid: Promptly remove any contamination from the skin, eyes, or clothing. Use a mild soap and water if required.